eazyplex® SARS-CoV-2
When it matters - eazyplex® SARS-CoV-2! The best combination of sensitivity, specificity and speed!
According to in-silico-analysis of the published sequences of SARS-CoV-2 variant B.1.1.529, the Omicron variant is reliably detected by eazyplex® SARS-CoV-2.
Use the optimal combination of the most innovative
Nucleic acid amplification technology and the user-friendly format of the eazyplex® system. With the eazyplex® SARS-CoV-2 you get the result without RNA extraction directly from UTM / VTM, eSwab medium or dry swabs in 25 minutes at the latest.
The portable device guides you through the test with its intuitive software. The sample control informs you about the smear quality and the automatically generated table provides clear results.
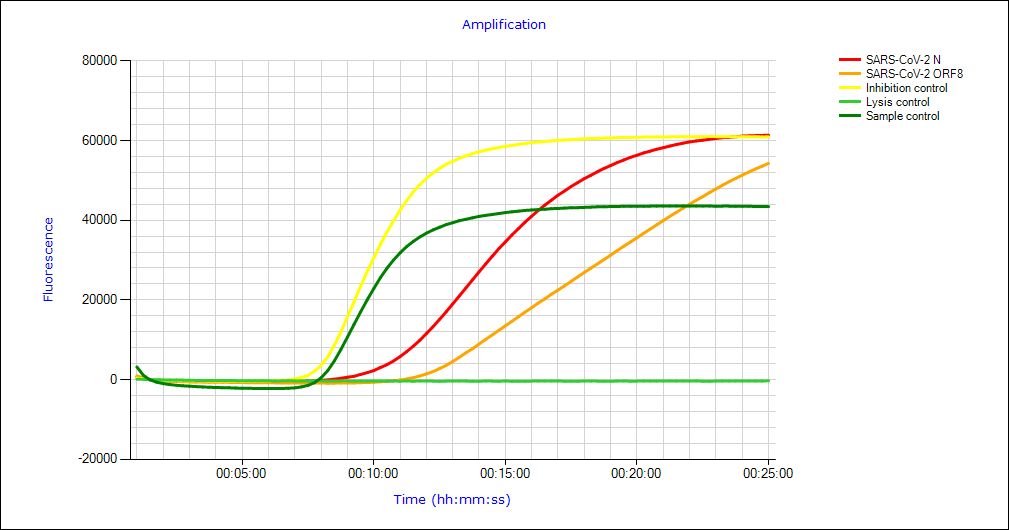
You receive maximum diagnostic reliability through the dual testing of the WHO-recommended target "N-gene" and the ORF8 gene.
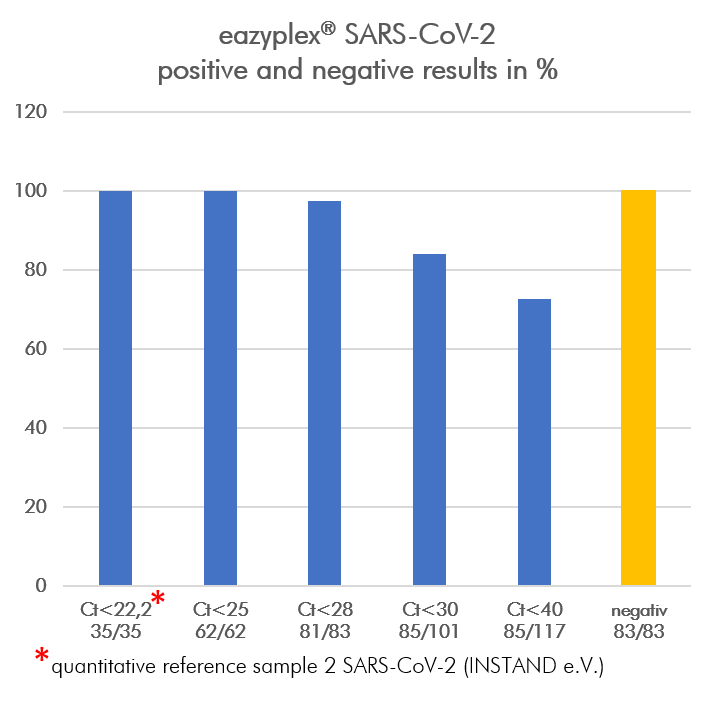
Sensitivity
In an evaluation study at Jena University Hospital (Institute for Medical Microbiology, Head of Study Prof. Dr. Rödel), oropharyngeal swabs from 200 different patients with the requirement "SARS-CoV-2" were taken between October 9 and November 13, 2020 and with the eazyplex® SARS-CoV-2 examined. The comparison was made with the NeuMoDx ™ SARS-CoV-2 Assay (NeuMoDx Molecular).
Specificity
The BLAST search (www.ncbi.nlm.nih.gov/blast/) shows that the selected primers contained in the eazyplex® SARS-CoV-2 detect SARS-CoV-2 (5000 sequences published on November 26, 2020 ).
SARS-CoV-2 ("Severe Acute Respiratory Syndrome Corona Virus 2")
In December 2019, the new corona virus was detected for the first time in the Chinese city of Wuhan. The disease caused by the virus was named by the WHO on February 11, 2020 as "COVID-19" or "Covid-19" as the abbreviation for "Corona Virus Disease 2019". And on the same day, the virus itself was classified by the CSG (Coronavirus Study Group) of the “International Committee on Taxonomy of Viruses (ICTV)” as SARS-CoV-2 for “severe acute respiratory syndrome coronavirus 2”. This name was chosen because of its genetic similarity to the 2003 SARS outbreak. Although the two viruses are genetically related, they are different. By March 10, 2020, over 100,000 COVID-19 cases had already been confirmed. On March 11, the WHO declared this outbreak a pandemic. As usual in coronaviruses, the virus genome consists of single-stranded RNA (ssRNA) with positive polarity. On February 16, 2020, SARS-CoV-2 had already carried out more than 40 complete genome analyzes. Two of the analyzes showed a close genetic relationship (89%) to the genome of the coronaviruses found in bats. Human-to-human transmission was deemed possible by the Chinese Health Commission in January. Both the US Center for Disease Control and Prevention and the Robert Koch Institute recommend a minimum distance of 1-2 m to reduce the infection rate. Although the virus has so far been detected in secretions of the nose and throat, sputum, stool, tear fluid and blood, it is assumed that SARS-CoV-2, just like other pathogens that cause respiratory diseases, is spread through droplet infection and aerosol in the air. All experts agree that rapid molecular biological diagnostics to contain the pandemic is very important. For fast and reliable virus diagnostics, AmplexDiagnostics GmbH offers the CE-IVD certified test system eazyplex® SARS-CoV-2.